Efficacy
Wirta, D. L., Walters, T. R., Flynn, W. J., Rathi, S., & Ianchulev, T. (2021). Mydriasis with micro-array print touch-free tropicamide-phenylephrine fixed combination MIST: pooled randomized Phase III trials. In Therapeutic Delivery (Vol. 12, Issue 3, pp. 201–214). Future Science Ltd. https://doi.org/10.4155/tde-2021-0011
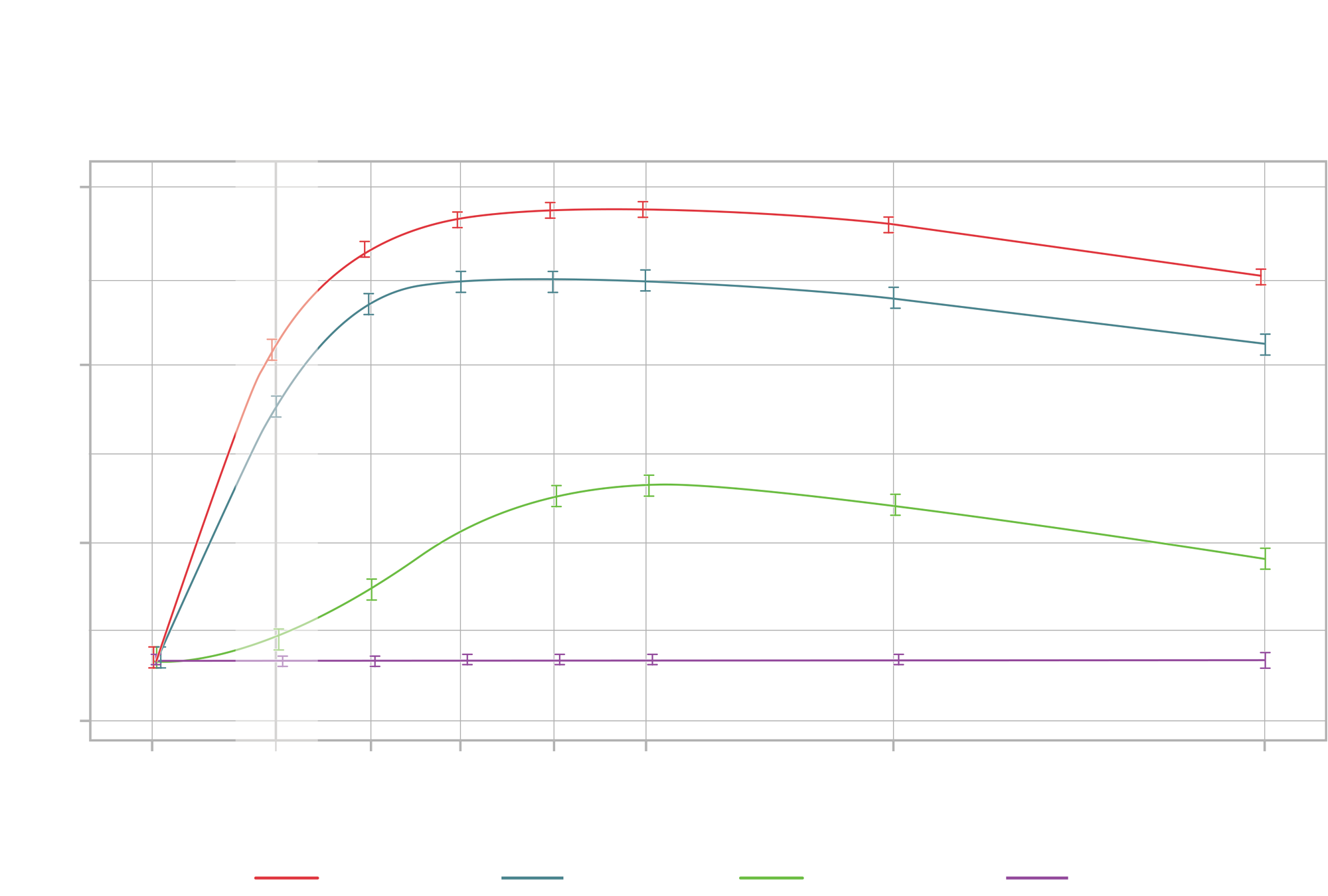
Pupil diameter by measurement time and treatment (pooled per-protocol population). Vertical bars show 95% CI for the mean at each point. Smooth curves are based on an 8 df GAM smooth through time, adjusting for baseline pupil diameter. Confidence intervals are not adjusted for correlation. GAM: Generalized additive models; PH: Phenylephrine; TR: Tropicamide; TR-PH: Fixed-combination tropicamide-phenylephrine.
- MydCombi introduces an ‘anesthetic-free’ dilation to reduce exposure to unnecessary agents, streamline routine ophthalmic care for patients, technicians and physicians thereby enhancing office efficiency, practice flow and patient experience
- The pooled results of these studies demonstrate that MydCombi produced clinically relevant and statistically superior pupil dilation compared with phenylephrine (PH), tropicamide (TR) and placebo
- The proportion of eyes that achieved a pupil diameter ≥6.0 mm at 35 min post dose was 93.3% with MydCombi compared with 78.2% with TR and 1.6% with PH.
Safety
- The rate of adverse events was low (4 out of 131 patients), and no systemic or serious adverse events were reported
- All ocular AEs were transient and resolved prior to study exit
- The design of MydCombi allows for high precision targeted administration of TR-PH (∼8 μl/dose) without use of prior anesthesia, resulting in improved patient comfort, lower drug exposure and significantly reduced overflow risk than with eye drops
- MydCombi has no protruding parts to touch the ocular surface, thus minimizing cross-contamination risk
Administering MydCombi
Important: Keep MydCombi dispenser upright during use to maintain dose volume.
1. Load drug solution
a. Hold MydCombi dispenser upright and firmly press Fill Button down until it stops, and Mist Light turns Blue.
b. Slowly release Fill Button, while counting to 3.
2. Align MydCombi with patient’s eye
a. Hold MydCombi dispenser with thumb over Mist Button, wrapping other fingers around base.
b. Bring MydCombi dispenser to patient’s eye with Mirror facing the eye.
The dispenser should be as close as patient’s nose. To prevent blinking, use your other hand to gently pull lower eyelid down or ask patient to pull their lid down.
c. Aim Mist Opening toward the center of eye.
d. Confirm Alignment Marks (on the Fill Button and the cartridge side) align with the center of eye
Ask patient to confirm when their eye is centered on the BLUE Mirror. If patient is having trouble centering their eye on the blue light, ask that they look up, then look at the BLUE Mirror.
3. Press Mist Button
a. Firmly press and release Mist Button.
The drug solution should gently wet the eye.
– Repeat Steps 1-3 if needed.
INDICATION
(pilocarpine hydrochloride ophthalmic solution) 1.25% is indicated for the treatment of presbyopia in adults.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
contraindicated in patients with known hypersensitivity to any ingredient in the formulation.
WARNINGS AND PRECAUTIONS
Miotics, including VUITY, may cause accommodative spasm. Patients should be advised not to drive or operate machinery if vision is not clear (e.g., blurred vision). In addition, patients may experience temporary dim or dark vision with miotics. Patients should be advised to exercise caution in night driving and other hazardous activities in poor illumination.
Rare cases of retinal detachment and retinal tear have been reported with miotics, including VUITY. Individuals with pre-existing retinal disease are at increased risk. Therefore, examination of the retina is advised in all patients prior to the initiation of therapy. Patients should be advised to seek immediate medical care with sudden onset of flashing lights, floaters, or vision loss.
it is not recommended to be used when iritis is present because adhesions (synechiae) may form between the iris and lens.
Contact lens wearers should be advised to remove their lenses prior to the instillation and to wait 10 minutes after dosing before reinserting their contact lenses.
To prevent eye injury or contamination, care should be taken to avoid touching the dispensing bottle to the eye or to any other surface.
ADVERSE REACTIONS
The most common adverse reactions (>5%) reported in clinical trials were headache, conjunctival hyperemia, and eye irritation.test